New Consolidator grantee at the Department of Biology
Associate Professor Mette Burmølle has received one of the highly acclaimed Consolidator grants from the European Research Council, ERC. The grant, as the name implies, helps researchers with promising scientific track records to consolidate their groups and continue their ground-breaking research. The proportion of female researchers being awarded the Consolidator grant is on the rise and for the grants received at UCPH the division is fifty-fifty.
 Mette Burmølle has received 15 mDKK in funding for the project BioMatrix, with the overall aim to identify the molecular mechanisms underpinning community intrinsic properties of bacterial communities. The project will run for 5 years, beginning next year.
Mette Burmølle has received 15 mDKK in funding for the project BioMatrix, with the overall aim to identify the molecular mechanisms underpinning community intrinsic properties of bacterial communities. The project will run for 5 years, beginning next year.
Background
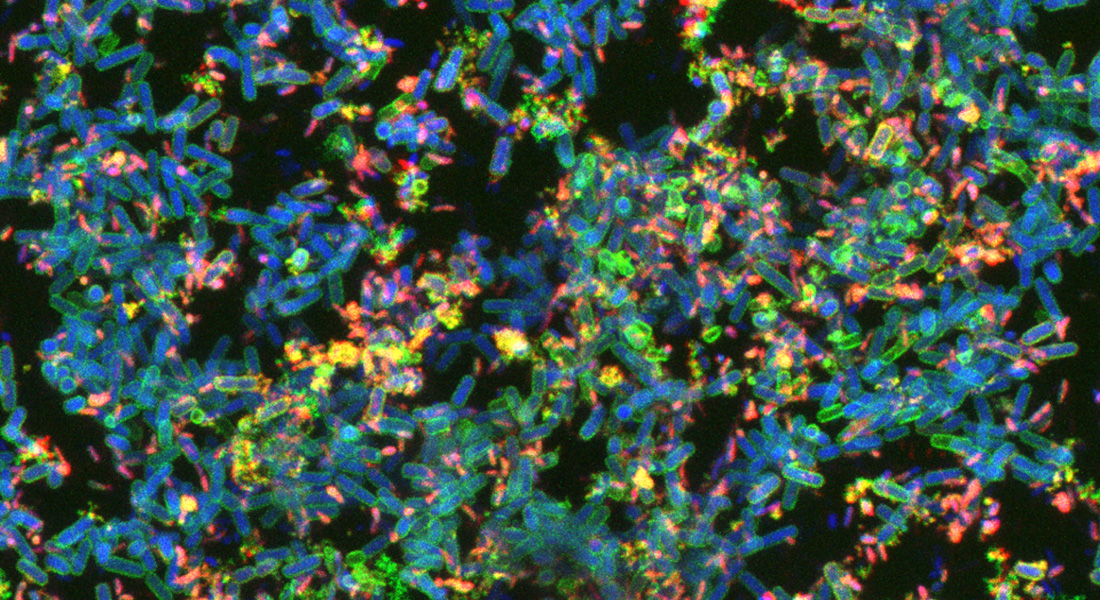
Bacteria in nature live in biofilms. These biofilms are found everywhere; on plant roots, on decomposing organic material, in aquatic sediments and much more. Biofilms are useful in biotechnological applications, but they are also problematic, as they form at unwanted sites in industrial settings and can lead to chronic infections in humans.
Biofilms are assemblages of bacteria embedded in a matrix, produced by the bacteria themselves. When different types of bacteria live in proximity in a biofilm, we often identify features of this microbial community, which neither of the bacteria possess individually; examples include enhanced tolerance towards stress and inhibitors, and new metabolic capabilities. The interactions and mechanisms causing such “community properties” are largely unknown.
BioMatrix
Mette Burmølle explains: ”We will examine the matrix of biofilms comprised of multiple types of bacteria. We aim at identifying matrix components that directly impact functionality and/or tolerance of biofilms in nature. New experimental models will be designed to better mimic the real life of biofilm bacteria in nature”.
Perspectives
BioMatrix will uncover fundamental mechanisms shaping bacterial communities and thereby close a primary knowledge gap in microbial ecology. Moreover, specific targets, facilitating efficient biofilm manipulation, are provided. This is useful in biotechnology for enhanced exploitation of bacterial community activity and in the development of novel and improved chronic infection treatment strategies.
Kontakt
Associate Professor Mette Burmølle
Section of Microbiology
Universitetsparken 15
2100 Copenhagen Ø
burmolle@bio.ku.dk
Phone: +45 35 33 01 19
Mobile: +45 40 22 00 69
Communications Officer
Sidsel Kretzschmer Henriksen
Department of Biology
sidsel.henriksen@bio.ku.dk
Phone: +45 35 33 41 47
