Signalling and Transport in Epithelia
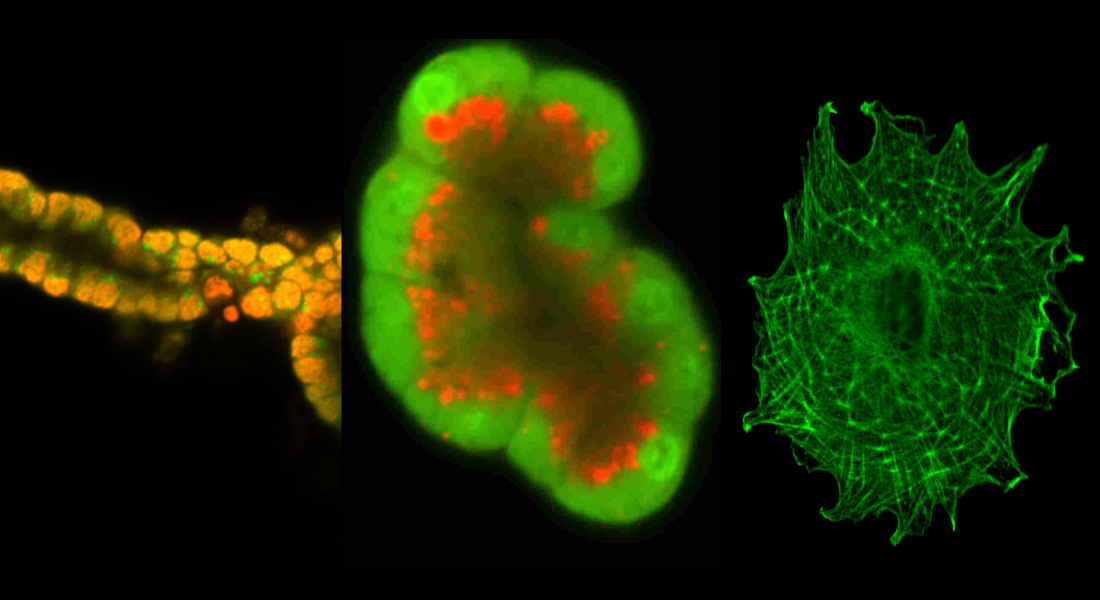 Our general aim is to understand the epithelial transport processes and their regulation at the molecular/cellular level and to integrate these into the normal physiological function of the chosen organs and the body. Pancreas is one of the organs of interest, and further goals are to understand how these transport processes play roles in pathophysiology of pancreas cancer, cystic fibrosis, pancreatitis and diabetes.
Our general aim is to understand the epithelial transport processes and their regulation at the molecular/cellular level and to integrate these into the normal physiological function of the chosen organs and the body. Pancreas is one of the organs of interest, and further goals are to understand how these transport processes play roles in pathophysiology of pancreas cancer, cystic fibrosis, pancreatitis and diabetes.
Current research includes projects on the role of extracellular ATP and purinergic signalling in regulation of ion transport, metabolism and behaviour of pancreatic and other cells in health and disease. We use many techniques, especially advanced bioimaging in our imaging center CAB.
The projects in our research group deal with fundamental processes in cell biology/physiology: cell to cell signalling via extracellular nucleotides/sides, acid/base transport and ion transport/homeostasis.
1. ATP Signals
ATP (adenosine 5´-triphosphate) is the molecule of life. Inside the cell it provides energy and participates in essential processes. Outside the cell it acts as a short-range and “evolutionary old” signalling molecule. This so called Purinergic Signalling involves a number of membrane-bound specific receptors and enzymes and ATP release mechanisms. Our aim is to understand interactions between cellular metabolism, ATP release and purinergic signalling in health and disease models such as diabetes, obesity and cancer. The project makes use of various cell models, including pancreatic cancer and stellate cells, beta-cells and adipocytes, and a number of techniques including various reporters and sensors imaged in living cells.
Funded by The Independent Research Fund Denmark (DFF)/Natural Sciences
2. Role of purinergic signalling in pancreatic cancer
Pancreatic cancer is a deadly cancer, and new disease markers and therapeutic targets are needed. To achieve this goal we need better understanding of cellular/molecular mechanisms involved in interactions between fibrogenic pancreatic stellate cells and cancer cells within the tumour. The aim of the project is to elucidate whether purinergic signalling, in particular genetic variants in P2X7R, could contribute to cross-talk between pancreatic stellate cells and pancreatic epithelial cells and contribute to development of pancreatic cancer.
Funded by The Independent Research Fund Denmark (DFF)/Medical Sciences and Talent Doctoral Programme (Marie Sklodowska-Curie COFUND) and collaboration with COST Action CA21130 - P2X receptors as a therapeutic opportunity (PRESTO)
3. pH and Ion Transport Proteins in pancreatic cancer
Ion channels and acid/base transporters underlie many of the hallmarks of cancer. In our EU consortium we are mapping and targeting the "transportome" and tumour pH microenvironment in pancreatic ductal adenocarcinoma (PDAC), which has one of the worst prognoses of all cancers. In our group we are investigating the role of proton transporters, ATP sensitive purinergic receptors, Calcium-activated Cl- and pH-sensitive K+ channels in PDAC development and behavior.
Funded by and collaborations in Marie Sklodowska-Curie Innovative Training Networks IonTraC and pHioniC
4. Acid/base transport in pancreas
Transport of acids and bases over cell membranes is essential for the function of every cell in the body. Some organs maintain body acid/base homeostasis, while others utilize acid/base transport for special purposes, such as digestion, and transport significant quantities of protons and bicarbonate. The overall goal of our project is to clarify why pancreas, which is secreting high concentrations of bicarbonate, expresses proton pumps and is sensitive to common proton pump inhibitors. This knowledge is used to study pancreatic diseases including pancreatic cancer and cystic fibrosis.
Funded by the Novo Nordisk Foundation and the Lundbeck Foundation
CA21130 - P2X receptors as a therapeutic opportunity (PRESTO)
pH and Ion Transport in Pancreatic Cancer - pHoniC

PhD schools
The PhD Graduate School of Molecular Biology and Physiology
PhD course on Advanced Live Cell Microscopy (CAB)
Cross Institutional Course BioImaging PhD course - DaMBIC
Master courses
Cellular and Integrative Physiology
Advanced Topics in Physiology
Bachelor courses
Human Physiology (Menneskets fysiologi)
Master and Bachelor projects
see under projects and Department of Biology Project database
The major part of our research is focused on visualization of physiological processes in living cells and organs. State-of-the-art Confocal laser scanning and multiphoton imaging microscope with FLIM and high throughput confocal station are part of a our facility in the "Center for Advanved Bioimaging" (https://www.cab.ku.dk/) at Nørre Campus and is run by our lab. Also electrophysiological recordings are used to monitor membrane transport processes in living cells.
Our laboratories are equipped for:
- Fluorescence optical methods - Leica SP 5XP MP with FLIM, Zeiss and Nikon microscopes equipped with EM-CCD and for live-cell imaging, Nikon Biostation, Luminometer - fluorometer for microtiter plate assays
- Electrophysiological recordings - microelectrode, patch-clamp, oocyte clamp and mini-Ussing chamber set-ups
- Tissue and cell preparations from animals and set up for whole animal (rat, gene manipulated mice) in vivo experiments and in vitro organ experiments
- Tissue culture of human cell lines (3D and organoids) and primary cell cultures of epithelia, pancreatic stellate cells, pancreatic islets, adipocytes and macrophages
- Cell and molecular biological methods including expression of proteins tagged with Fluorophores, FRET, immunolocalization techniques, migration, survival assays etc
Contact
Signalling and Transport
in Epithelia
Cell Biology and Physiology
Universitetsparken 13
DK-2100 Copenhagen Ø, Denmark
Professor Ivana Novak
Email: inovak@bio.ku.dk
Phone: +45 3533 0275

